|
The starting material, only primary amides can be produced by this process. Treatment of nitriles with a solution of HCl in ethanol furnishes an imidateĮster which is hydrolyzed in aqueous acid to the amide. DirectĪcid or base hydrolysis of a nitrile usually requires fairly severe conditionsĪnd often does not stop at the amide stage but goes on the carboxylic acid. Urea by-product from the carbodiimide can be separated and removed from theĪre also available from nitriles, which have the same oxidation level. The activated species need not be isolated. The functional or R groups of amino acids are the groups that define the chemistry of proteins. The advantage of thisĪpproach is that the acid is activated to a reactive electrophile in situ so An activated acylating agent is produced which reacts with theĪmine present in the mixture to produce an amide. benzamide, N, N -dimethylmethanesulfonamide, secondary amides (see imides ), tertiary amides, phenylphosphonamidic acid. Very common one is dicyclohexylcarbodiimide (DCC), although many others haveīeen developed]. Compounds having one, two or three acyl groups on a given nitrogen are generically included and may be designated as primary, secondary and tertiary amides, respectively, e.g. In this method the carboxylic acid is treated with a carbodiimide [a This method was originally developed for creating the amide bonds in The car-boxylate ion, which is an even poorer electrophile than the carboxylicĪre increasingly being used to promote the conversion of car-boxylic acids toĪmides. Many nucleophiles are also basic, they can react with carboxylic acids to give Amide general formula We now know that amides contain a carbonyl group, CO, bonded to an amine group, -NH 2. Check out Amines and The Carbonyl Group for more information about these two functional groups. This consists of a carbonyl group bonded to an amine group. functional group has a carbonyl group joined to a nitrogen atom from ammonia or an amine. Amides are organic molecules with the amide functional group, -CONH2. Sufficiently electrophilic to react with most nucleophiles. The functional group of an amine is a nitrogen atom with a lone pair of electrons and with one, two, or three alkyl or aryl groups attached. Transformations of carboxylic acids because carboxylic acids themselves are not This approach, formation of an acid chloride is required to activate the car-bonyl However,Įxcess amine or another base is required to neutralize the equivalent of HCl 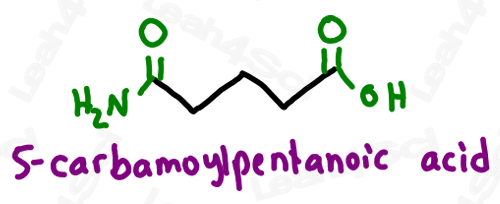
This remains an excellent and efficient method. Substitution process replaces the chloride with an amine without a change in Tradi-tional method of preparation of amides is to react the corresponding acid The tradi-tional method of preparation of amides is to react the corresponding acid chloride with an amine.Īre usually obtained from carboxylic acids or their derivatives. Chapter: Organic Chemistry : Functional Group SynthesisĪmides are usually obtained from carboxylic acids or their derivatives.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
